The Gas Laws

In this section, we discuss:
- The Importance of Gas Laws
- Boyle’s Law
- Henry’s Law
- Dalton’s Law
- Charles’ and Amontons’ Laws
- The General Gas Law
- Why is it important for divers to know and understand the gas laws?
- What is Boyle’s Law?
- What is Henry’s Law?
- What is Dalton’s Law?
- What are Charles’ Law and Amontons’ Law?
- What is the General Gas Law?
The Importance of Gas Laws
In regard to gases and diving, there are three factors that come into play: pressure, temperature and volume. Gas laws define the relationships between these three factors. A change in one of these three, interrelated factors in a gas results in changes in the other factors. Divers should know and understand the applications of relevant gas laws so that they can understand how individual changes in pressure, temperature or volume affect the other factors and their physiology.

For example, divers should know how the reduction of pressure during an ascent effects the volume of air in their lungs. As the pressure decreases, the volume of air in the lungs increases as the air expands. If divers hold their breath, they could suffer a lung expansion injury. Rather than testing this in person, divers rely on the gas laws to predict and describe the interrelation of volume and pressure.
Basically, the gas laws are a set of rules that predict interaction of pressure, temperature, and volume of a gas.
Boyle’s Law
Boyle’s Law states that for any gas at a constant temperature, the volume is inversely proportional to the pressure. Simply stated, as pressure increases, volume decreases and as pressure decreases, volume increases.
Boyle’s Law can be expressed using this formula:
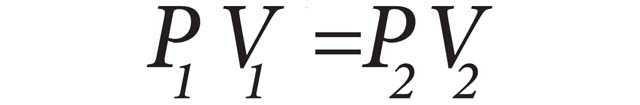
P1 is the initial pressure, V1 is the initial volume, P2 is the final pressure and V2 is the final volume.

Remember this diagram from your entry-level scuba course? It not only shows the relationship between pressure and volume, but also the relationship between pressure and density. Where pressure and volume are inversely related, pressure and density are directly related. This explains why gas consumption increases with depth.
Henry’s Law
Henry’s Law states that at any given temperature, the weight of a gas dissolved by a liquid is directly proportional to the partial pressure of the gas upon the liquid. Simply stated, as you dive deeper, more gas will dissolve in your body tissues.
Henry’s law can be put into mathematical terms (at constant temperature) as:
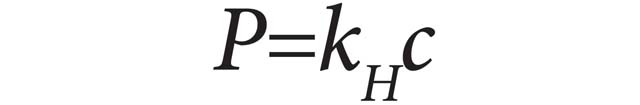
P is the partial pressure of the gaseous solute above the solution, C is the concentration of the dissolved gas and kH is a constant with the dimensions of pressure divided by concentration. The constant, known as the Henry's law constant, depends on the solute, the solvent and the temperature.
You can use Henry’s Law to understand the relationship between pressure (depth) and gas absorption. Henry’s Law provides a framework for explaining why dive tables and computers give you less bottom time the deeper you go. Since your body absorbs more gas as it goes deeper, the more gas you have to release as you surface. Consequently, you need to ascend at 10 m/30 ft per minute or slower to manage off-gassing.
Even though Henry’s Law refers to liquid, it applies to us because our bodies are mostly water.
Dalton’s Law
Dalton’s Law states that the total pressure exerted by a mixture of a gas is equal to the sum of the pressures of each of the different gases making up the mixture, with each gas acting as if it alone was present and occupied the total volume. Simply stated, a gas mixture is the sum of its parts.
In any gas mixture, the portion of the total pressure of the gas that a single gas exerts is call the partial pressure of that gas. It can be expressed mathematically as:
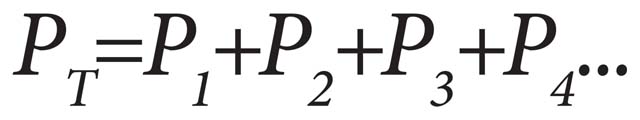
You can use the following formula to calculate the partial pressure of oxygen (PO2) in a gas mixture at any given depth, provided you know its concentration in the gas mixture (FO2).
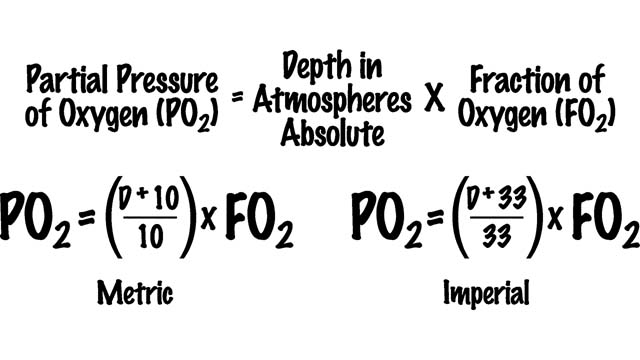
By substituting other values, you can also use this formula to calculate the partial pressure of nitrogen or other gases present.
Charles’ and Amontons’ Laws
Charles’ Law states that at a constant pressure, the volume and temperature of a gas are directly proportional. Simply stated, if temperature of a gas increases, then the volume increases. Charles’ Law can be expressed mathematically as:
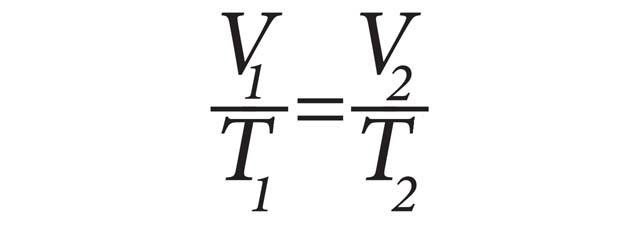
V1 is the initial volume, T1 is the initial temperature, V2 is the final volume, and T2 is the final temperature.
Amontons’ Law states that at a constant volume, the pressure and temperature of a gas are directly proportional. Simply stated if the temperature of a gas increases, then the pressure increases. Amontons’ Law can be expressed mathematically as:
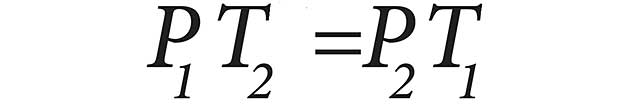
P1 is the initial pressure, T1 is the initial temperature, P2 is the final pressure and T2 is the final temperature.
Remember that Boyle’s Law defines the relationship between pressure and volume, with temperature as a constant. However, in the real world, temperature does not remain constant and changes in temperature significantly affect volume and pressure.
While Charles’ Law and Amontons’ Law separately address temperature’s relationship with volume and pressure respectively, it should be apparent that if any of the three factors, pressure, volume or temperature, change then the other at least one other factor or both will be affected. This why we need a law that predicts the behavior of a gas when all three factors change. This is the General Gas Law.
The General Gas Law
The General Gas Law combines three gas laws to predict the behavior of a quantity of gas when either temperature, pressure or volume changes. The General Gas Law can be expressed mathematically as:
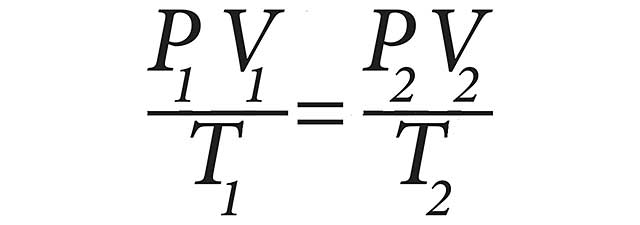
V1 is the initial volume, P1 is the initial pressure, T1 is the initial temperature, V2 is the final volume, P2 is the final pressure and T2 is the final temperature.
Keep in mind that you can have only one unknown factor. Also, you can simplify the formula if one of the factors remains constant. For instance, if temperature remained unchanged, you could remove it from the formula by canceling it out on both side. In this case, the formula would then revert from the General Gas Law back to Boyle’s Law.
Key Points to Remember
- Because gas laws define the relationships between pressure, temperature and volume of a gas, divers should know and understand the applications of certain gas laws, so that they can understand how individual changes in pressure, temperature or volume affect the other factors and their physiology.
- For any gas at a constant temperature, the volume of a gas is inversely proportional to the pressure. In contrast, the density of a gas is directly proportional to pressure. (Boyle’s Law)
- At any given temperature, the weight of a gas dissolved by a liquid is directly proportional to the partial pressure of the gas upon the liquid. (Henry’s Law)
- The total pressure exerted by a mixture of a gas is equal to the sum of the pressures of each of the different gases making up the mixture, with each gas acting as if it alone was present and occupied the total volume. (Dalton’s Law)
- Charles’ Law states that at a constant pressure, the volume and temperature of a gas are directly proportional. Amontons’ Law states that at a constant volume, the pressure and temperature of a gas are directly proportional.
- The General Gas Law combines Boyle’s Law, Charles’ Law, and Amontons’ Law to predict the behavior of a quantity of gas when either temperature, pressure or volume changes.
Learn More on Wikipedia
Important!

You will need to refer directly to the Master Diver eBook in order to answer the assessment questions appearing on the next page. If you have not already done so, download the book now and have it open in Adobe Acrobat while you tackle each of the problems.
